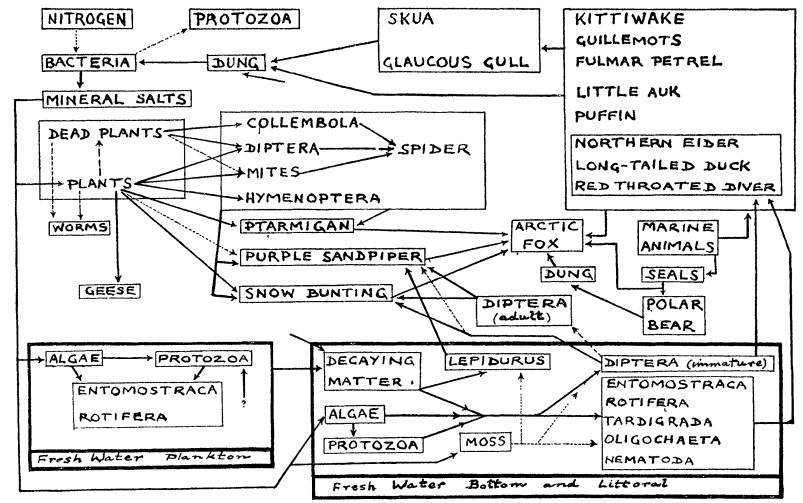
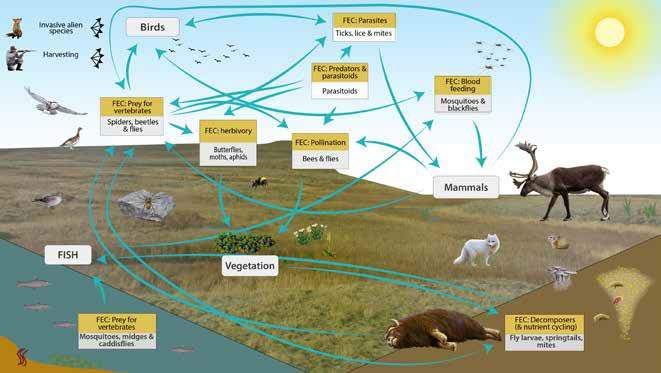
The group invertebrate contains a great diversity of taxa and groups; from hard-bodied insects to soft bodied organisms such as worms.
Given this diversity it should not come as a surprise that they have a great diversity of ecosystem functions as well. To attempt to describe these functions in a manageable system to be employed with monitoring strategies the Arctic Terrestrial Biodiversity Monitoring Plan, Terrestrial Expert Monitoring Group forming part of the Cicumpolar Biodiversity Monitoring Programme (Christensen et al. 2013) devised a system of Functional Ecosystem Components that could be used as bins to classify species. While constructed to enable monitoring the FECs also describe the key ecosystem functions performed by the invertebrate communities. While the Arctic Terrestrial Biodiversity Monitoring Plan 2013 identifed five FECs, Gillespie et al 2020 identified a further, that of Predators (e.g. parasitoids).
Text from Arctic Terrestrial Biodiversity Monitoring Plan (Christiensen et al. 2013)
- Blood-feeding invertebrates. Flies are among the most commonly encountered animals in the Arctic, and biting flies (Diptera; several groups) are notorious for their blood-feeding behaviours. All Arctic biting flies have aquatic larval stages but occupy terrestrial habitat as adults, and include black flies (Simuliidae), mosquitoes (Culicidae), ‘deer flies’ or ‘moose flies’ (Tabanidae), and others. Adult females blood-feed for the successful development of their eggs, and hence are nuisance pests and can introduce pathogens and parasites. Biting flies also play critical roles in pollination, especially in northern systems. Biting flies are a primary reason for the initiation of the Northern Insect Survey and are among the most well-known of the northern insects. Furthermore, biting fly harassment of wildlife may be increasing, and with additional compounded effects of climate change, may further increase negative impacts on populations. For these reasons, they must be part of an Arctic monitoring plan.
- Pollinators. The co-dependence of flowering plants and their invertebrate pollinators requires that both biotic groups be included in integrated monitoring efforts and that attributes such as spatial structure (distributions), diversity, abundance, and phenology be considered as much as possible. In the Arctic, groups such as Hymenoptera, and Diptera may be the most important and feasible to monitor. Because of the familiarity of many species (e.g., bumble bees) and as some protocols can be adapted for ease of use (e.g., observing flower visitation rates) community-based monitoring can be an invaluable component of programs.
- Prey species for vertebrates. Many arthropods and invertebrates are exploited by birds and small mammals, and in the Arctic, monitoring is proposed for spiders (Araneae), some flies (Tipulidae) and for Lepidoptera, for feasibility. For many migratory avian breeders, one major driver of their extensive journeys is to exploit the rich sources of invertebrate prey that emerge in time to coincide with the high energy demands of reproduction and for feeding offspring. Climate change may be causing drought conditions in some tundra regions, which over time may affect aquatic larval stages of insect prey. The shifting phenology of prey and predators, and mismatched reproductive periods, could impact avian populations and other predators of invertebrates.
- Decomposers and nutrient-cyclers. Soil and tundra meso- and micro-fauna perform critical ecosystem functions including breaking down larger organic particles and facilitating decomposition, facilitating the movement of energy and nutrients along food webs from microbes to small prey, burrowing and creating air spaces in soil that benefit vegetation and other species, and serving as predators, fungivores, detritivores, and prey. Monitoring can be practical for at least some groups, and where possible, samples of other species should be archived so that these can be analysed as capacity permits in the future. Under the current plan, soil invertebrates that should be monitored include soil mesofauna (springtails, mites, and Enchytraeids), fungi, earthworms, and microfauna and microorganisms (some requiring DNA-based analyses).
- Herbivores. As many arthropod taxa are phytophagous (Lepidoptera, Aphidae, Coleoptera, Acari, etc.), herbivores are dependent and influence their host plants and thus vegetation in the Arctic. They are expected to respond quickly to changes in vegetation and monitoring programs should include them as they could signal important changes in ecosystems. However, selecting key taxa for inclusion is challenging as taxonomic capacity for some groups is virtually non-existent, whereas other taxa are known well, especially in their adult forms (e.g., butterflies).
- Predators. (from Gillespie et al. 2020). As an intermediate trophic level of Arctic food webs, this group is critical for community dynamics and is likely to be more responsive to changes in lower trophic levels than their vertebrate predators. Predatory arthropods make up a large proportion of the prey for vertebrates FEC. While a predator FEC may be a more appropriate functional group for many of these species there are also many uncertainties around such a category. For example, many parasitoids perform other functions (e.g., pollination), and apart from spiders, the larval diets of many species are unknown or vary between regions and habitats.
Functions of the terrestrial and freshwater invertebrate fauna as described by Functional Ecosystem Components (FEC) State of the Arctic Terrestrial Biodiversity Report (Aronsson et al. 2021 and Ambio Gillespie et al 2020a).
Steve Coulson
The multi-functionality of Arctic arthropods. The chord diagram indicates the number of species in each FEC for the North Atlantic (circular outline) and the overlap between the five CBMP FECs and our recommended ‘‘Predators’’ FEC. Link width is indicative of the number of species presumed to link the various FECs. Note that 34 species found in 3 or more FECs are not depicted, so the total number of species is slightly inflated.
- Christensen, T., J. Payne, M. Doyle, G. Ibarguchi, J. Taylor, N.M. Schmidt, M. Gill, M. Svoboda, M. Aronsson, C. Behe, C. Buddle, C. Cuyler, A.M. Fosaa, A.D Fox, S. Heiðmarsson, P. Henning Krogh, J. Madsen, D. McLennan, J. Nymand, C. Rosa, J. Salmela, R. Shuchman, M. Soloviev, and M. Wedege. 2013. The Arctic Terrestrial Biodiversity Monitoring Plan. CAFF Monitoring Series Report Nr. 7. CAFF International Secretariat. Akureyri, Iceland. ISBN 978-9935-431-26-4
- Gillespie, M.K.A., Alfredsson, M., Barrio, I.C., Bowden, J., Convey, P., Culler, L.E., Coulson, S.J., Krögh, P.H., Koltz, A.M., Koponen, S., Loboda, S., Marusik, Y., Sandstrøm, J.P., Sikes, D. & Høye, T.T. (2020) Status and trends of Arctic terrestrial arthropod diversity of the North Atlantic region. Ambio 49;718–731